AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Orbital diagrams for all elements1/11/2024 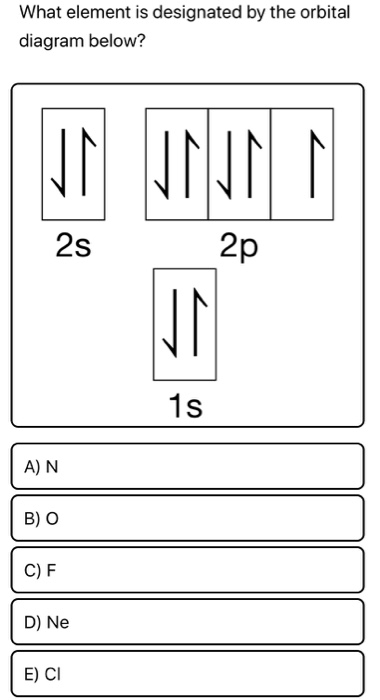
In keeping with convention, the unpaired electrons are drawn as "spin-up", which gives (Figure 1). We will now construct the ground-state electron configuration and orbital diagram for a selection of atoms in the first and second periods of the periodic table. Hund's rule also stipulates that all of the unpaired electrons must have the same spin. The traditional orbital elements are the six Keplerian elements, after Johannes Kepler and his laws of planetary motion. Substance 2: 7) For the second substance, draw an orbital diagram like the one in the video. Therefore, two p orbital get one electron and one will have two electrons. According to Hund’s rule, all orbitals will be singly occupied before any is doubly occupied. Construct a molecular orbital diagram for each element based on the location of the valence. That leaves 4 electrons, which must be placed in the 2p orbitals. All covalent bonds are either sigma () or pi () bonds. We can show the distribution of electrons by using box diagrams, where each box represents an orbital and the arrows within the boxes represent the electrons in. 
To draw the orbital diagram, begin with the following observations: the first two electrons will pair up in the 1s orbital the next two electrons will pair up in the 2s orbital. The electron configuration can be written as 1s 22s 22p 4. The Aufbau principle predicts that the 4s orbital is always filled before the 3d orbitals, but this is actually not true for most elementsFrom Sc on, the 3d orbitals are actually lower in energy than the 4s orbital, which means that electrons enter the 3d orbitals first. There are 118 elements in the periodic table. Identify the noble gas that comes BEFORE the element you are writing the electron. \): Carbon and OxygenĬonsider the electron configuration for carbon atoms: 1s 22s 22p 2: The two 2s electrons will occupy the same orbital, whereas the two 2p electrons will be in different orbital (and aligned the same direction) in accordance with Hund's rule.Ĭonsider also the electron configuration of oxygen. All electrons in singly occupied orbitals must have the same spin.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
